














|
 |
| |
|
|
| |
 E-mail to a friend
E-mail to a friend  Printer friendly
Printer friendly |
|
| |
|
|
 |
When
neurologist Allan Levey arrived at Emory in 1991,
there wasn’t much he could do for his patients with Alzheimer’s
disease. He would explain the mysterious beta amyloid plaques
in the brain that clogged synapses between neurons, and he could
prepare patients and families for the next step in the patient’s
deteriorating condition as well as take care of coexisting medical
problems. But he watched too many patients simply waste away with
agonizing slowness, bankrupting families financially and emotionally
over the course of many years. And he became tired of it.
“For so long, Alzheimer’s
has been seen as a hopeless disease,” says Levey, chair
of the Department of Neurology. “That is no longer the case.
We are actually at a place in time where we can offer treatments
that have been proven to work.”
Levey collaborated on a landmark
article published in the June 9, 2005, issue of the New England
Journal of Medicine, concluding that the drug Aricept can
delay the progression of Alzheimer’s for as long as three
years if administered early in the course of the disease. “This
is the most important clinical trial done to date in Alzheimer’s
disease because it marks the very first time that we have delayed
its onset,” Levey says. “It is a treatment that may
work best for people at very high genetic risk. We showed an outcome
that established that these people should be treated very early,
and while we still have a long way to go, it was the first time
we proved we could intervene in Alzheimer’s disease and
make a difference.”
 uring the past 15 years, Levey has helped link a vast network
of Alzheimer’s research, clinical, and education projects
at Emory. Having expanded exponentially in quantity and funding,
these efforts form the crux of the Alzheimer’s Disease Research
Center (ADRC), a recent designation granted by the National Institute
of Aging (NIA). Along with the five-year, $7.4 million grant,
the relevance of this designation is enormous, says Levey, who
directs the center. The grant will attract more research dollars
to Emory’s already formidable neurodegenerative disease
efforts. The designation distinguishes Emory as one of the foremost
research and clinical centers for Alzheimer’s disease in
the country. The center also is supported by the Georgia Research
Alliance, a private nonprofit partnership among statewide university
and industry leaders and government, which will allow recruitment
of an eminent scientist and investments in cutting-edge technologies
and equipment.
uring the past 15 years, Levey has helped link a vast network
of Alzheimer’s research, clinical, and education projects
at Emory. Having expanded exponentially in quantity and funding,
these efforts form the crux of the Alzheimer’s Disease Research
Center (ADRC), a recent designation granted by the National Institute
of Aging (NIA). Along with the five-year, $7.4 million grant,
the relevance of this designation is enormous, says Levey, who
directs the center. The grant will attract more research dollars
to Emory’s already formidable neurodegenerative disease
efforts. The designation distinguishes Emory as one of the foremost
research and clinical centers for Alzheimer’s disease in
the country. The center also is supported by the Georgia Research
Alliance, a private nonprofit partnership among statewide university
and industry leaders and government, which will allow recruitment
of an eminent scientist and investments in cutting-edge technologies
and equipment.
Boosted by basic science research
in genetics and proteomics and advances in clinical care, research
has yielded an explosion of new knowledge about Alzheimer’s.
Technological advances in brain imaging and data management also
have sped discoveries.
Creighton Phelps, program director
of the ADRCs at the NIA, says that only the very best university
research programs receive this designation. “Emory was one
of only two new centers funded by the NIA in 2005,” Phelps
says, “and the process was intensely competitive.”
Emory joins a network of 32 NIA
centers nationwide dedicated to understanding the development,
progression, diagnosis, and treatment of Alzheimer’s and
related neurodegenerative diseases. The emphasis of the Emory
center is to catch the disease as early as possible and halt its
progress. |
 |
| |
|
|
 |
 |
 |
| |
|
|
 |
The
importance of early intervention |
|
 |
One
tack Emory researchers are taking is to understand the role of
mild cognitive impairment as a precursor to Alzheimer’s
disease. “Not all people with mild cognitive impairment
will develop Alzheimer’s disease, but it is a risk factor,”
says Levey.
For their clinical studies, the
researchers are enrolling a combination of healthy volunteers
and those with mild cognitive impairment from the patient population
at Wesley Woods Center and Grady Memorial Hospital. “We
can have the most impact if we can find ways to identify the disease
early and intervene before too much damage is done,” Levey
says.
Despite the emphasis on early intervention
to halt Alzheimer’s disease, early diagnosis is difficult
because the disease shares so many symptoms with other neurologic
disorders such as Parkinson’s disease, mini-strokes, and
vascular disease. Enter Stuart Zola, director of the Yerkes National
Primate Research Center and co-director of the Emory ADRC. Zola
is applying his research in memory formation, consolidation, and
retrieval to Alzheimer’s questions. One of the first scientists
to develop a model of human amnesia in nonhuman primates that
identified brain structures critical to memory function, he has
helped scientists understand memory loss in humans resulting from
head trauma and neurodegenerative diseases. His work with monkeys
and its application to humans has resulted in the development
of new diagnostic behavioral tools for diagnosing humans with
mild cognitive impairment and with early-stage Alzheimer’s
disease. It is crucial, according to Levey.
“We now have a much better,
more specific definition of Alzheimer’s disease, and we
can diagnose it with much greater confidence and at much earlier
stages than before.” |
 |
| |
|
|
 |
 |
 |
| |
|
|
 |
Not-so-sweet
dreams |
|
 |
Emory
researchers are also busy on another front: exploring the role
of sleep and neurologic dysfunction in Alzheimer’s. Donald
Bliwise, Emory professor of neurology and director of the Program
in Sleep, Aging, and Chronobiology, says many medical problems
associated with aging—such as insulin resistance, hypertension,
hypothyroidism, and low testosterone—may contribute to the
progression of Alzheimer’s disease and cognitive decline.
Sleep apnea, which affects 20% of
the geriatric population, causes chronic oxygen deprivation (hypoxia)
in the brain. Falling blood oxygen levels from periods of depressed
breathing during sleep also stress the heart. A huge burst of
autonomic sympathetic nervous system activity follows to awaken
those in the throes of a sleep apnea episode. Apparently, once
this nervous system sensitivity is set off, it’s hard to
stop, and as a result, more stress hormones are released both
day and night. Research has shown that sleep apnea causes both
hypertension and insulin resistance. The cumulative effects of
all these factors may play a role in neurologic decline, says
Bliwise.

“In our clinical studies,
we’re finding that many Alzheimer’s patients have
a long history of sleep apnea,” says Bliwise. “When
you talk to patients and their caregivers in the clinic, you hear
so much about snoring and gasping at night. The brain is a target
organ for sleep apnea. There has been more of an emphasis on the
cardiovascular effects of sleep apnea, but little attention has
been paid to its relationship to Alzheimer’s disease. We’re
charting new ground.”
In fact, in addition to family history
of the disease, identifying those with sleep apnea may be an important
way to flag people at higher risk for Alzheimer’s, says
Levey. |
 |
| |
|
|
 |
 |
 |
| |
|
|
 |
A
visual diagnosis |
|
 |
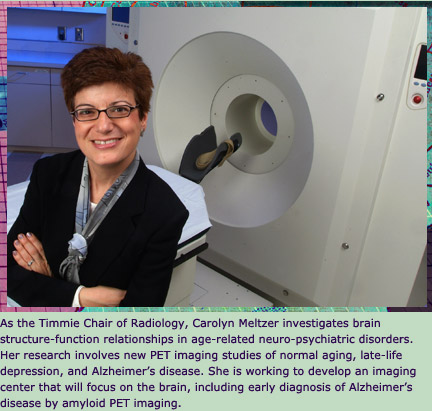
In
the past, an Alzheimer’s diagnosis was little more than
educated guesswork. Now, Emory radiologists are developing tools
that offer a more definitive diagnosis via a picture of an Alzheimer’s
brain.
Mark Goodman, professor of radiology
and director of the PET (positron emission tomography) Imaging
Center at Emory, says PET images offer a look at brain function
in living patients, allowing researchers to visually track the
brain’s metabolism of glucose and oxygen. After many years
of work, he has discovered a revolutionary pharmaceutical compound
that when used with 3-D PET scans can highlight beta amyloid plaques
in the living brain.
“This agent gives us the ability
to image the cognitive area of the brain—the frontal cortex—and
see the beta amyloid plaques in a living person,” says Goodman.
“Previously, we could see these plaques only after patients
had died, too late to do any good. The ability to see and measure
the severity of the plaques changes everything in Alzheimer’s
diagnosis. It takes out the guesswork.”
This compound can highlight plaques
in several stages of development and is the first shown to be
clinically practical. It is a radioactive molecule that has an
affinity for beta amyloid plaques and chemically binds to them.
When injected into the bloodstream, it has good delivery to the
brain and allows for high-contrast images of amyloid plaques.
“Our invention is the first
that could be widely distributed to clinics and hospitals,”
Goodman says. “It has a half-life of eight hours. The only
other such agent known to work has a half-life of 20 minutes,
making it impractical for use outside the lab.”
The center is conducting a study
using brain imaging and biomarker tests together with measures
of memory, thinking, and daily functioning to see if an earlier
diagnosis of Alzheimer’s can be made. The research participants
include a control group, a group with mild cognitive impairment,
and a group with Alzheimer’s disease. The two-year study
should lead to a much faster process for better diagnostic tools
and for testing medications that are targeted at the molecules
that cause the disease. |
 |
| |
|
|
 |
 |
 |
| |
|
|
 |
Plucking
proteins from plaques |
|
 |
Genetics
assistant professor Junmin Peng gestures proudly at a large wooden
box taking up most of the hallway outside his lab. Inside is a
new tool, the very latest technology in mass spectrometry, and
Peng can’t wait to unpack it.
“Just as a scale measures
weight, the mass spectrometer measures the mass of very small
molecules, like proteins,” Peng says. Matching a protein’s
mass to the map of the human genome provides an accurate identification
of the protein.
“Without the database of the
human genome, we can only measure the mass,” Peng says.
“But with the human genome information, we can use the mass
to identify the protein and quantify it.”

Peng uses this tool to study the
most basic proteins in beta amyloid plaques in brains of Alzheimer’s
patients postmortem. Peng and neuropathologists at Emory use a
laser scanning microscope to locate amyloid plaques and then pluck
out a sample. The mass spectrometer measures the mass of the proteins
comprising the plaques. Then, a powerful computing center in the
Department of Human Genetics—32 processors in all—matches
the mass to the proper protein, making a positive I.D.
Proteomics, or the study of proteins,
is opening new windows to understanding the development of Alzheimer’s
disease, whether it’s caused by the absence of certain normal
proteins in early Alzheimer’s patients or the buildup of
too many.
Sequencing of the human genome has
propelled the field of proteomics, offering opportunities to apply
it to diseases such as Alzheimer’s. “There was an
explosion of information from the human genome map, which has
a huge impact on our research,” Peng says. “It provides
a framework like a whole database of the potential proteins present
in humans. Mass spec-based proteomics is a young field, but it’s
advancing very rapidly. We are on the cutting edge by applying
it to Alzheimer’s and making a difference for real people—not
just learning about molecules.” |
 |
| |
|
|
 |
 |
 |
| |
|
|
 |
It
all comes back to the patient |
|
 |
Past
the fancy science and evolving high-tech inroads to a mysterious
disease, making a difference for people is still the central focus
of the Emory researchers and physicians. Every Monday, Levey spends
the day at Wesley Woods, seeing inpatients and holding clinics
for outpatients and their families. Many of his patients are involved
in one of the many clinical studies that fall under the umbrella
of the Emory ADRC.
Caring for Alzheimer’s patients
must also involve an entire family unit. “It is a familial
disease with a strong genetic origin,” Levey says. “Family
ties are very important, both as risk factors and in caring for
patients.”
Alzheimer’s disease takes
a huge toll on caregivers. The need for emotional support and
education is crucial to keeping patients out of nursing homes
and in the best possible surroundings. “We recognize that
the proper support must be in place for this to work,” Levey
says. “Here at Wesley Woods, we’re providing that
for families.”
Beyond educating families, reaching
out to the larger community is another goal. For example, the
clinical center at Grady Memorial Hospital treats mostly African-American
patients with Alzheimer’s, examining racial disparities
in treatment and outcomes of the disease as well as cultural differences
in how families cope. According to Levey, early dementia sometimes
involves different factors in African-American patients, such
as a higher prevalence of high blood pressure and diabetes.
Data from all these clinical and
scientific studies are collected and sent to the NIA annually.
There the numbers are combined with those from all the other research
centers around the country in nothing less than a full-scale national
assault on what was once considered a hopeless disease. Now prevention
and even a possible cure for Alzheimer’s disease are no
longer just a dream. “We will do amazing things with this
disease in my lifetime,” says Levey. “We’re
so close I can practically taste it.” |
 |
| |
|
|
| |
|
|
|
|
|





